Keiichiro Susuki, MD, PhD
Research Statement
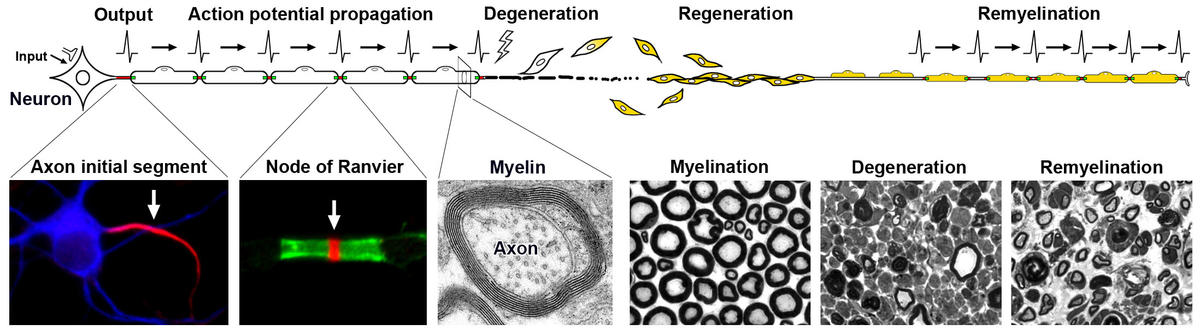
As a neuroscientist with a clinical background, the mission of my research is to enhance our understanding of nervous system development and injury to improve care for patients with neurological diseases. Action potentials are generated at the axon initial segment, a specialized functional domain within neurons that lies between the soma and the axon proper. Rapid and efficient propagation of the action potentials is ensured by the neuron-glia interactions forming myelin (a multilamellar structure to insulate axons) and the node of Ranvier (a short gap between two adjacent myelin sheaths). Dysfunction/disruption of these structures is a major factor leading to neurological symptoms in a broad range of neurological diseases. However, few treatments are available to directly protect myelinated nerves or facilitate nerve regeneration, because of a limited knowledge of the mechanisms regulating assembly or disruption of these structures. My long-term research goals are to 1) determine the molecular/cellular mechanisms of formation, maintenance, injury, and repair of myelinated nerve fibers, and 2) develop novel therapeutic approaches for currently intractable neurological diseases. As a member in the translational Neuroscience Institute, my research currently focuses on the glial cell cytoskeletons as well as the molecular complex at the axon initial segment and nodes of Ranvier, utilizing genetically modified mice, rodent nerve injury models, and a mouse cortical neuron culture system. We have assembled a talented team of undergraduate and graduate students, and postdoctoral fellows within the lab.
Education History:
- Postdoctoral: Department of Neuroscience, Baylor College of Medicine, TX (2007-2008)
- Postdoctoral: Department of Neuroscience, University of Connecticut Health Center, CT (2005-2007)
- PhD: Yokohama City University School of Medicine, Japan (2001)
- MD: Yokohama City University School of Medicine, Japan (1994)
Funding:
- NINDS R01 NS107398 (8/1/2019-7/31/2024), Supplement (1/1/2020-7/31/2021)
- NINDS R03 NS112981 (9/15/2019-8/31/2021)
- NINDS R56 NS107398 (8/15/2018-7/31/2019)
Funding awarded to trainees:
- NIA/NINDS NRSA F30 NS124237 to Jennae N. Shelby (8/1/2021-7/31/2025)
- Original Work Grant (Graduate Student Assembly, Wright State University) to Duc Van Minh Nguyen (4/26/2021-12/31/2021)
- Medical Student Research Grant (Boonshoft School of Medicine, Wright State University) to Fatima Z. Bensabeur (10/20/2019-3/31/2021)
Teaching:
- NEU4500/PN6550 Neuron-Glia Biology and Disease: Course director
- PN7750/BMS8750 Neuroscience and Physiology: Action potential propagation, Excitable axonal domains I & II
- ANT7310/BMS9030 Human Neurobiology: Cells of the CNS and PNS, Thalamus, Cerebral cortex, Cerebellum, Cranial nerves
- WrightQ: Facilitator of case discussions
Students Advised:
- Jennae N. Shelby, MD/PhD candidate in Biomedical Sciences, Thesis Advisor: August 2019-present.
- John A. Miller, PhD candidate in Biomedical Sciences, Thesis Advisor: May 2019-present.
- Leonid M. Yermakov, MD/PhD in Biomedical Sciences, Thesis Advisor: June 2016-PhD awarded March 2019.
- Duc Van Minh Nguyen, MS candidate in Physiology and Neuroscience, Thesis Advisor: August 2019-present.
- Domenica E. Drouet, MS in Anatomy, Thesis Advisor: August 2017-MS awarded May 2019.
- Fatima Z. Bensabeur, MD candidate, Research Advisor: July 2019-graduated May 2021.
- Josef K. Steinbrunner, Neuroscience Undergraduate Honors Program, Thesis Advisor: June 2019-graduated May 2020.
- Duc Van Minh Nguyen, Biological Sciences Undergraduate Honors Program, Thesis Advisor: Sept 2017-graduated Dec 2018.
Publications
Selected publications (complete list: https://www.ncbi.nlm.nih.gov/myncbi/1f_lolqYSIck_/bibliography/public/)
[Research articles]
- Griggs RB, Nguyen DVM, Yermakov LM, Jaber JM, Shelby JN, Steinbrunner JK, Miller JA, Gonzalez-Islas C, Wenner P, Susuki K (2021). The type 2 diabetes factor methylglyoxal mediates axon initial segment shortening and alters neuronal function at the cellular and network levels. eNeuro 8:ENEURO.0201-21.2021
- Chang K-J, Agrawal I, Vainshtein A, Ho WY, Xin W, Tucker-Kellogg G, Susuki K, Peles E, Ling S-C, Chan JR (2021). TDP-43 maximizes nerve conduction velocity by repressing a cryptic exon for paranodal junction assembly in Schwann cells. eLife 10:e64456
- Eshed-Eisenbach Y, Devaux J, Vainshtein A, Golani O, Lee S-J, Feinberg K, Sukhanov N, Greenspan DS, Susuki K, Rasband MN, Peles E (2020). Precise spatiotemporal control of nodal Na+ channel clustering by bone morphogenetic protein-1 /tolloid-like proteinases. Neuron 106:806-815.
- Yermakov LM, Griggs RB, Drouet DE, Sugimoto C, Williams MT, Vorhees CV, Susuki K (2019). Impairment of cognitive flexibility in type 2 diabetic db/db mice. Behav Brain Res 371:111978.
- Griggs RB, Santos DF, Laird DE, Doolen S, Donahue RR, Wessel CR, Fu W, Sinha GP, Wang P, Zhou J, Brings S, Fleming T, Nawroth PP, Susuki K, Taylor BK (2019). Methylglyoxal and a spinal TRPA1-AC1-Epac cascade facilitate pain in the db/db mouse model of type 2 diabetes. Neurobiol Dis 127:76-86.
- Yermakov LM, Drouet DE, Griggs RB, Elased KM, Susuki K (2018). Type 2 diabetes leads to axon initial segment shortening in db/db mice. Front Cell Neurosci 12:146.
- Susuki K, Zollinger DR, Chang K-J, Zhang C, Huang CY, Tsai C-R, Galiano MR, Liu Y, Benusa SD, Yermakov LM, Griggs RB, Dupree JL, Rasband MN (2018). Glial βII spectrin contributes to paranode formation and maintenance. J Neurosci 38:6063-6075. (Cover art)
- Griggs RB, Yermakov LM, Drouet DE, Nguyen DVM, Susuki K (2018). Methylglyoxal disrupts paranodal axoglial junctions via calpain activation. ASN Neuro 10:1759091418766175.
- Otani Y, Yermakov LM, Dupree JL, Susuki K (2017). Chronic peripheral nerve compression disrupts paranodal axoglial junctions. Muscle Nerve 55:544-554.
- Yoo S-W, Motary MG, Susuki K, Prendergast J, Mountney A, Hurtado A, Schnaar RL (2015). Sialylation regulates brain structure and function. FASEB J 29:3040-3053.
- Chang K-J, Zollinger DR, Susuki K, Sherman DL, Makara MA, Brophy PJ, Cooper EC, Bennett V, Mohler PJ, Rasband MN (2014). Glial ankyrins facilitate paranodal axoglial junction assembly. Nat Neurosci 17:1673-1681.
- Zhang C, Susuki K, Zollinger DR, Dupree J, Rasband MN (2013). Membrane domain organization of myelinated axons requires betaII spectrin. J Cell Biol 203:437-443.
- Susuki K, Chang K-J, Zollinger DR, Liu Y, Ogawa Y, Eshed-Eisenbach Y, Dours-Zimmermann MT, Oses-Prieto J, Burlingame AL, Seidenbecher C, Zimmermann DR, Oohashi T, Peles E, Rasband MN (2013). Three mechanisms assemble central nervous system nodes of Ranvier. Neuron 78:469-482.
- Susuki K, Yuki N, Schafer DP, Hirata K, Zhang G, Funakoshi K, Rasband MN (2012). Dysfunction of nodes of Ranvier: a mechanism for anti-ganglioside antibody-mediated neuropathies. Exp Neurol 233:534-542.
- Susuki K, Raphael AR, Ogawa Y, Stankewich MC, Peles E, Talbot WS, Rasband MN (2011). Schwann cell spectrins modulate peripheral nerve myelination. Proc Natl Acad Sci USA 108:8009-8014.
- Susuki K, Rasband MN, Tohyama K, Koibuchi K, Okamoto S, Funakoshi K, Hirata K, Baba H, Yuki N (2007). Anti-GM1 antibodies cause complement-mediated disruption of sodium channel clusters in peripheral motor nerve fibers. J Neurosci 27:3956-3967.
- Susuki K, Baba H, Tohyama K, Kanai K, Kuwabara S, Hirata K, Furukawa K, Furukawa K, Rasband MN, Yuki N (2007). Gangliosides contribute to stability of paranodal junctions and ion channel clusters in myelinated nerve fibers. Glia 55:746-757.
[Review articles]
- Yermakov LM, Hong LA, Drouet DE, Griggs RB, Susuki K (2019). Functional domains in myelinated axons. Adv Exp Med Biol 1190:65-83.
- Griggs RB, Yermakov LM, Susuki K (2017). Formation and disruption of functional domains in myelinated CNS axons. Neurosci Res 116:77-87.
- Susuki K (2016). Node of Ranvier disruption in Guillain-Barré syndrome. Clin Exp Neuroimmunol 7:324-329.
- Susuki K, Otani Y, Rasband MN (2016). Submembranous cytoskeletons stabilize nodes of Ranvier. Exp Neurol 283:446-451.
- Susuki K, Kuba H (2016). Activity-dependent regulation of excitable axonal domains. J Physiol Sci 66:99-104.
- Susuki K (2013). Node of Ranvier disruption as a cause of neurological diseases. ASN Neuro 5:e00118.
- Susuki K (2010). Myelin: A specialized membrane for cell communication. Nature Education 3:59.
- Susuki K, Rasband MN (2008). Molecular mechanisms of node of Ranvier formation. Curr Opin Cell Biol 20:616-623.